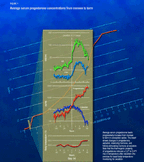
Progesterone in early pregnancy: By Laurie Jane McKenzie, MD, and John E. Buster, MD When during the first trimester is it valuable to measure levels of this hormone and when is it a waste of time? Will giving your patients progesterone supplements help prevent miscarriage? Are they safe? Progesterone has been aptly named the "hormone of pregnancy," because in preparing the endometrium for embryo implantation and facilitating endometrial development, it's critical to the very survival of a pregnancy. In addition, this key hormone inhibits the rejection of T cell-mediated tissue and also decreases myometrial activity and sensitivity throughout pregnancy. 1 More than a quarter century ago human studies dramatically demonstrated the importance of the corpus luteum's production of progesterone in pregnancy through classic experiments in which they surgically removed this ovarian "yellow body" in early pregnancy. When they performed this luteectomy before 7 weeks' gestational age, the pregnancy almost always led to miscarriage. They found that administering progesterone could prevent miscarriage in women who had a luteectomy. 2 More than 25 years before that experiment, progesterone was first extracted from the corpus luteum, characterized as a steroid, and later purified. Of course, we now refer to any steroid that exhibits progesterone-like activity as a progestin or progestogen. All progestins/progestogens transform the endometrium following estrogen-induced proliferation. Progesterone is produced predominantly by the ovaries, and in small measure by the adrenal glands, and at a rate of less than 1 mg/day before ovulation. After ovulation and corpus luteum formation, this increases to 20 to 30 mg/day. Then during pregnancy, it climbs dramatically, reaching 200 to 400 mg/day at term. Until approximately 7 weeks' gestation, increased progesterone production is dependent on the corpus luteum and thereafter on the placenta, the so-called "luteal-placental shift" which occurs between 7 and 10 weeks (Table 1 and Figure 1).
Graph of midcycle hormone levels is adapted with permission from: Hoff JD, Quigley ME, Yen SS. Hormonal dynamics at midcycle: a reevaluation. J Clin Endocrinol Metabol . 1983;57:792-796. Copyright 1983, The Endocrine Society. PLEASE NOTE: If you are using Internet Explorer version 6 or higher, after clicking to enlarge the graphic, click the picture icon that appears in the lower right-hand corner of the graphic. Our goal here is to discuss the practical value of measuring progesterone in the first trimester in four different situations and then to weigh the merits of giving therapeutic supplementary progesterone for indications like preventing threatened miscarriage. We'll also look at various formulations and routes of administration. When should you measure progesterone levels? Progesterone levels can confirm ovulation, help to uncover an ectopic pregnancy, and predict viability. The hormone's role in diagnosing luteal phase defect, however, is controversial. Documenting ovulation. Progesterone makes neurons within the hypothalamus fire faster. 3 This in turn increases a woman's set point temperature, resulting in the sustained rise in core temperature found during the luteal phase. Monitoring of basal body temperature draws upon this thermogenic property of progesterone to confirm that a woman has ovulated. For most women the morning basal temperature before rising is less than 98°F before they ovulate and above 98°F after ovulation, with an overall change ranging from 0.1° to 0.6°F. Ovulation occurs about 8 hours (median time) after plasma progesterone begins to rise. 4 In most situations, obtaining a single midluteal serum progesterone measurement should confirm ovulation. Usually, a plasma value above 3 ng/mL suggests a secretory endometrium and is presumed to indicate ovulation. 5 Determining a normal pregnancy. Investigators have extensively evaluated the importance of measuring progesterone very early in pregnancy. Compared to women going to term, women who spontaneously abort in the first trimester have lower levels of progesterone. 6 First-trimester values above 25 ng/mL suggest a normal intrauterine pregnancy 98% of the time, while pregnancies with values below 5 ng/mL are almost always nonviable. You may find this of limited usefulness, however, because many patients fall into the mid-range of these values. Screening for ectopic pregnancy. Because it's well-established that patients with ectopic pregnancies have low serum progesterone, some researchers propose taking a single progesterone measurement as a minimally invasive screen for ectopic pregnancy. 7,8 Most algorithms use a cutoff concentration ranging from 20 to 25 ng/mL to indicate with high specificity but poor sensitivity the presence of a viable pregnancy, thereby effectively excluding an ectopic pregnancy. However, other algorithms use a cut-off as low a 5 ng/mL, a value that maximizes sensitivity but leads to more false-positive—and falsely reassuring—results for the presence of a viable pregnancy. A recent meta-analysis of 26 studies showed that progesterone values were helpful—up to a point: They did well at discriminating between pregnancy failure and viable intrauterine pregnancy. Unfortunately, though, they could not discriminate between ectopic and nonectopic pregnancy. 9 Thus, a single serum progesterone measurement can at least identify patients at risk for an ectopic pregnancy, but cannot discriminate sufficiently to definitively diagnose ectopic pregnancy. Patients determined by this screen to be at risk can then undergo transvag- inal sonography and serum hCG measurements to specify the diagnosis. 10 Diagnosing luteal phase defect. Inconsistencies in diagnosis and management have led to much controversy about how defects in human luteal function affect reproduction. Luteal phase defects refer to below normal levels of progesterone produced by the corpus luteum, causing delayed endometrial development. Researchers have long suspected that this ovulatory disorder contributes to infertility and recurrent spontaneous abortion. The accepted gold standard for diagnosis remains an endometrial biopsy in which a developmental delay of more than 2 days is present in at least two cycles. Despite strict criteria for diagnosing a luteal phase defect, pathologists' interpretations of endometrial biopsies vary widely. In one out of three patients whose biopsies were read by five different pathologists, differences in interpretation were significant enough to alter clinical management. 11 Even the same pathologists re- reading coded endometrial biopsy slides a second time disagreed with 75% of their own original diagnoses. 12 A luteal phase "defect" appears to be widespread in healthy fertile women—and in fact, a recent definitive study actually found the same number of luteal phase defects in fertile and infertile women. In this prospective, randomized multicenter trial, a single pathologist reviewed all 619 endometrial biopsies. The study found that a high proportion of both fertile (49.4%) and infertile (43.2%) women had an out-of-phase biopsy during the window of implantation. The test failed to provide useful information and calls into question the value of endometrial biopsy as a screening tool for evaluating the infertile patient. 13 Experts also disagree about whether it pays to try to correct the luteal phase defect when treating women's reproductive problems. Therapy has not been proven effective, with one meta-analysis showing no beneficial effect with progesterone supplementation. 14 It may be that a luteal phase defect simply reflects the release of an abnormal oocyte. While without question, low progesterone and retarded endometrial maturation occur during abnormal ovulation, a luteal phase defect is probably the result rather than the cause of the abnormal ovulation. In other words, serum progesterone measurements have no value in the diagnosis of luteal phase defects. When should you give progesterone supplements? Besides the common practice of using progesterone supplementation during assisted reproduction, we'll also discuss whether the hormone is indicated for inducing periods in anovulatory women, preventing miscarriage, or inducing ovulation. Finally, we'll touch on the issue of whether progesterone therapy later in pregnancy is likely to prevent preterm births. To induce menses in anovulatory women? Giving progestins to an anovulatory patient can indeed bring about a withdrawal bleed. A typical example is a woman with polycystic ovary syndrome who ovulates irregularly. Assuming her cycle is longer than 35 days, she'll achieve a regular withdrawal bleed by taking either medroxyprogesterone acetate (MPA) 5 to 10 mg daily, or 200 to 400 mg of micronized progesterone daily—for 10 days each month. We recommend you do a pregnancy test before giving the progestin. To prevent threatened miscarriage ? Although many clinicians have widely prescribed progesterone to women with threatened abortion in the past, no well-designed trials have ever substantiated the initial optimistic reports of its effectiveness. The best designed studies all show progesterone is no more effective than placebo in preventing miscarriage and advise against progestin supplementation. 15-17 A meta-analysis of 15 randomized trials reached a similar conclusion. 18 Although some practitioners persist in this therapy, most have abandoned it due to lack of effectiveness and possible complications. Recently, for example, a 30-year-old woman developed a retinal artery occlusion after taking high-dose IM progestogen for 2 weeks to prevent a threatened miscarriage. 19 Clearly, there is no role for progesterone supplements in managing a threatened miscarriage, particularly as progesterone measurements and transvaginal ultrasound virtually eliminate any psychological need for them. To induce ovulation? Although no data support the practice of giving progesterone to patients undergoing ovulation induction with gonadotropins and intrauterine insemination, it's still common. We do recommend administering progesterone, however, if a gonadotropin-releasing hormone (GnRH) agonist is given before administering gonadotropins, because the corpus luteum will require additional exogenous support. But the effectiveness of this support in non-GnRH agonist cycles warrants clinical study. For IVF? Progesterone is commonly used after embryo transfer during in vitro fertilization (IVF). Specialists often give GnRH agonists or antagonists to prevent premature LH surges in women undergoing IVF. Unfortunately, the agonists inhibit the corpus lutea in the cycles, leading many practitioners to supplement with progesterone in the luteal phase in an attempt to compensate for this "iatrogenic luteal phase defect." Their aim in supplementing is to assist a corpus luteum that may have been compromised during ovulation induction or oocyte retrieval. 20 Another proposed use of progesterone during IVF cycles is to combat the effects of supraphysiologic estradiol levels on the endometrium. Although initial data suggested that elevated estradiol levels would adversely affect embryo implantation, a recent study found otherwise. Comparing 410 women undergoing IVF-ET (embryo transfer) with 181 egg donors and their recipients, it found no difference in implantation, pregnancy, and delivery rates between the two groups as a function of estradiol levels. 21 Despite progesterone's widespread use, there's no consensus on an ideal agent or route of administration. Both progesterone and human chorionic gonadotropin (hCG) help to provide routine luteal support in IVF. 22,23 The greatest drawback to using hCG for IVF is the risk of ovarian hyperstimulation, and therefore, most clinicians choose progesterone, with or without estrogen. Although most protocols advocate progesterone supplementation throughout the first trimester of pregnancy, recent studies suggest that this may not be necessary once pregnancy is detected. 24,25 Administering progesterone before embryo retrieval, however, seems to result in a lower clinical pregnancy rate than when it's given after oocyte retrieval. 26 The two most common routes for administering progesterone supplements for IVF are IM injection and vaginal (gels, capsules, suppositories), but they're also available in an oral micronized form (see, " Progesterone formulations and routes of administration ,"). The oral formulations, however, seem to provide inferior luteal support, according to a recent review of the literature on luteal phase support with IVF. 23 Pregnancy rates following IM versus vaginal routes were comparable, despite higher serum progesterone levels after IM injection. 20 Progesterone often plays a role in frozen embryo transfers, as well. By giving estrogen and progesterone, exogenous cycle control can be achieved, which facilitates timing of the transfer, particularly in anovulatory women. More than 1,500 thaw-and-transfer cycles from a single institution have yielded similar implantation and pregnancy rates with natural versus estrogen/progesterone-supplemented cycles. 27 To prevent preterm delivery? As for giving progesterone later than the first trimester, investigators are currently evaluating its effectiveness in preventing preterm delivery. A recent randomized placebo-controlled trial compared administration of 17 alpha-hydroxyprogesterone caproate progesterone versus placebo to prevent preterm birth in high-risk women. 28 The study was terminated early when the results showed significant protection against recurrent preterm birth. Questions certainly remain about optimal dosage and formulation, and its applicability to patients with other high-risk obstetric factors, such as multiple gestations, cervical length, or positive results for fetal fibronectin. 29 Further studies are ongoing. Is progesterone a teratogen? In 1977, the FDA recommended restricting use of progestogens in early pregnancy, based on early studies that showed that MPA might cause defects in cardiac development, CNS effects, amelia, and masculinization of female fetuses. 30-32 Other investigators found a dose-related induction of cleft palate by excessive dosing of MPA in rabbits, but not in mice or rats. 33 Researchers retrospectively evaluated a human study of 390 mothers of infants with congenital heart disease to assess exposure to estrogen and progesterone in early pregnancy. 34 The study found a small positive association (prevalence ratio estimate of 1.5, 90%, CI 1.0–2.1). Both the poor confidence interval and recall basis limit the interpretation of this study. Moreover, these early studies were often contradictory and inconclusive, chiefly due to a lack of uniformity of study material and different methodologies. One researcher suggested that MPA or a metabolite might bind to specific glucocorticoid receptors to inhibit or alter normal steroidal function in embryo-fetal development. 35 Many subsequent and better-designed studies, however, have failed to show any such harmful effect. One of the larger studies evaluated 2,754 infants born to women who experienced vaginal bleeding during their first trimester. During the first 3 months of pregnancy, 1,608 women received progestin (mostly MPA) therapy, and the control group comprised 1,146 infants of untreated mothers. There was no difference in the malformation rate between the two groups (120/1,000 vs. 123.9/1,000). 36 This study was followed by an evaluation of 1,016 pregnancies in which 449 women received MPA from the 5th to 7th week of pregnancy. Once again, no differences were found with regards to congenital abnormalities in the treated versus untreated group. Based on such data, progesterone doesn't seem to put an embryo at risk, nor is it likely to cause an abnormal fetus to be retained that might otherwise abort. The American College of Obstetricians and Gynecologists objected to the initial FDA labeling of progestogens, stating "there are no data to indicate that the use of progesterone causes any teratogenic effects...and is disturbing to infertil-ity patients taking progesterone." In April 1999 the FDA published its intent to revoke its regulation requiring manufacturers to warn patients of the possibility of birth defects associated with progestational drugs. There have been no suc-cessful lawsuits associated with the use of oral progestins in the first trimester of pregnancy (Upjohn - Pharmacia, oral communication, September 2003). Conclusions Progesterone is essential to the survival of an early pregnancy and its levels within the bloodstream rise predictably during gestation. In certain circumstances, we can apply what we've learned about normal concentrations of this hormone during the first trimester. We can measure progesterone levels to document ovulation, predict pregnancy viability, and help identify ectopic pregnancies. The role of progesterone in diagnosing luteal phase defect is controversial, however. As for first-trimester progesterone therapy, there's a lack of evidence for supplementation in most clinical settings, especially to prevent threatened miscarriage. However, various formulations of progesterone supplements are widely administered for assisted reproduction. Although progesterone supplementation given after the first trimester is beyond the scope of this paper, a recent RCT has again raised hopes that it can prevent preterm delivery, but further studies are ongoing. Progesterone formulations and routes of administration Progesterone is the only natural progestin with any significant biologic activity. Natural progesterone is available in two oral forms: crystalline progesterone (which is poorly absorbed when swallowed), and a micronized form, which is better absorbed orally. Vaginal and intramuscular preparations are also available. Synthetic progestins, which are more potent, are divided into either progesterone-like or testosterone-like compounds. Those structurally similar to progesterone include 17 alpha-hydroxyprogesterone acetate, MPA, megestrol acetate, and cyproterone acetate. Those structurally similar to testosterone are then further divided into two classes: those related to norethindrone (norethindrone acetate, ethynodiol acetate, norethynodrel, and norethindrone enanthate); and those related to levonorgestrel (desogestrel, norgestimate, and gestodene, often referred to as "new generation" progestins.). 1 Oral progesterone. This route may seem appealing due to its ease of administration, but poor efficacy and many side effects (sedation, flushing, nausea, and fluid retention) limit its use. 2,3 High variability in oral absorption and rapid metabolism by the liver cause progesterone to degrade to its 5-alpha and 5-beta reduced metabolites. 4,5 The better oral absorption of micronized progesterone (Table A) is achieved through modification with an acetoxy group at the 17 position, an alteration exemplified by MPA and megestrol acetate. Endometrial biopsies show that oral progesterone is ineffective in producing an in-phase endometrium in women with ovarian failure. 6
IM progesterone. Despite the wide use of IM progesterone in oil—particularly in ART programs—patients find this formulation inconvenient and uncomfortable. It can also lead to allergic reactions, inflammation, and even sterile abscess formation. 7 Vaginal routes. The disadvantages of gel and suppository vaginal preparations are that they can cause vaginal irritation and discharge, and require several daily doses. Serum progesterone levels are significantly higher after IM administration versus vaginal gel; however, endometrial concentrations are markedly higher with the vaginal preparation due to lymphatic infiltration. 8
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||